 Where C p is the heat capacity at constant pressure and α is the coefficient of (cubic) thermal expansion. There are expressions in terms of more familiar variables such as temperature and pressure: This energy storage on the atomic level includes energy associated with electron orbital states, nuclear spin, and binding forces in the nucleus.Įnthalpy is represented by the symbol H, and the change in enthalpy in a process is H 2 – H 1. On the other hand, energy can be stored in the chemical bonds between the atoms that make up the molecules. It is the thermodynamic quantity equivalent to the total heat content of a system. It is the energy contained within the system, excluding the kinetic energy of motion of the system as a whole and the potential energy of the system as a whole due to external force fields. The SI unit of enthalpy is the joule (J). For a variable-pressure process, the difference in enthalpy is not quite as obvious.Įnthalpy in Extensive Units Extensive and intensive properties of medium in the pressurizer.Įnthalpy is an extensive quantity, it depends on the size of the system, or on the amount of substance it contains. At constant pressure, the enthalpy change equals the energy transferred from the environment through heating (Q = H 2 – H 1) or work other than expansion work. It is due to the fact, it simplifies the description of energy transfer. It is so useful that it is tabulated in the steam tables along with specific volume and specific internal energy. The enthalpy is the preferred expression of system energy changes in many chemical, biological, and physical measurements at constant pressure. 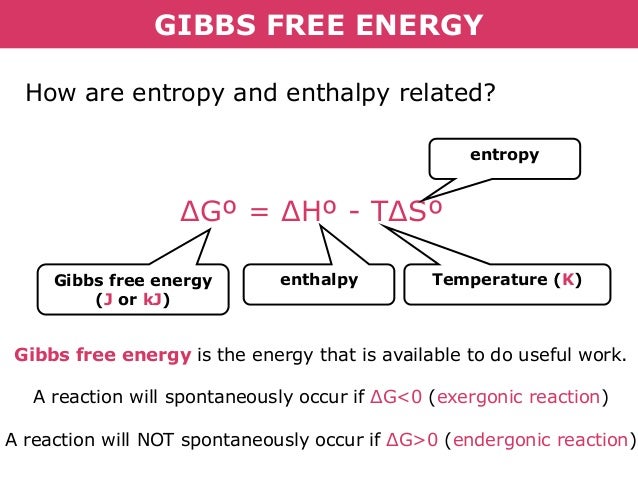
In many thermodynamic analyses the sum of the internal energy U and the product of pressure p and volume V appears, therefore it is convenient to give the combination a name, enthalpy, and a distinct symbol, H. The enthalpy is defined to be the sum of the internal energy E plus the product of the pressure p and volume V. In thermodynamics, the enthalpy is the measure of energy in a thermodynamic system. What is Enthalpy Four common thermodynamic potentials. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
